SYNVISC® Dosing and Administration
Review the dosing schedule for the SYNVISC family of viscosupplements to see which treatment option is right for your patients. Plus, get step-by-step injection instructions and watch a video demonstration about a technique for injecting SYNVISC/Synvisc-One.
Dosing ►
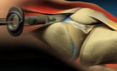
Injection Demonstration Video
Learn about needle placement and how to inject Synvisc-One.®
MAT-US-2005369-v2.0-01/2024
Resources for Your Patients
MAT-US-2106837-v1.0-07/2021
Indication
SYNVISC® (hylan G-F 20) and SYNVISC-ONE® (hylan G-F 20) are indicated for the treatment of pain in osteoarthritis (OA) of the knee in patients who have failed to respond adequately to conservative nonpharmacologic therapy and simple analgesics, e.g., acetaminophen.
IMPORTANT SAFETY INFORMATION
Important Safety Information
SYNVISC and SYNVISC-ONE are contraindicated in patients with known hypersensitivity to hyaluronan products or patients with infections in or around the target knee. Hypersensitivity reactions including anaphylactic reaction, anaphylactoid reaction, anaphylactic shock and angioedema have been reported for both SYNVISC and SYNVISC-ONE.
Do not concomitantly use disinfectants containing quaternary ammonium salts for skin preparation because hyaluronan can precipitate in their presence. Do not inject SYNVISC or SYNVISC-ONE extra-articularly, into the synovial tissues, into the fat pad or joint capsule, or intravascularly. Some cases of skin necrosis have been reported after intra-articular use of hyaluronic acid. Patients should be instructed to contact their treating physician if signs of skin disorder (such as change of color or open sores) appear.
The safety and efficacy of SYNVISC-ONE in locations other than the knee, or for conditions other than osteoarthritis, or in combination with other intra-articular injectables, or in severely inflamed knee joints have not been established. Use caution when injecting SYNVISC or SYNVISC-ONE in patients allergic to avian proteins, feathers, or egg products; who have evidence of lymphatic or venous stasis in the leg to be treated; or who have severe inflammation in the knee to be treated. Remove any synovial fluid or effusion before injecting SYNVISC or SYNVISC-ONE. Strict adherence to aseptic technique must be followed to avoid joint infection. The safety and effectiveness of SYNVISC and SYNVISC-ONE have not been established in children (≤21 years old) or in pregnant or lactating women. Patients should be advised to avoid strenuous or prolonged weight-bearing activities for approximately 48 hours after treatment.
For SYNVISC
In clinical trials, the most commonly reported adverse events were transient pain, swelling, and/or joint effusion in the injected knee. The following reported adverse events are among those that may occur in association with intra-articular injections, including SYNVISC: arthralgia, joint stiffness, joint effusion, joint swelling, joint warmth, injection site pain, arthritis, arthropathy, and gait disturbance.
View the Complete Prescribing Information for SYNVISC
For SYNVISC-ONE
The most commonly reported adverse events were arthralgia, arthritis, arthropathy, injection site pain, and joint effusion. The following reported adverse events are among those that may occur in association with intra-articular injections, including SYNVISC-ONE: arthralgia, joint stiffness, joint effusion, joint swelling, joint warmth, injection site pain, arthritis, arthropathy, and gait disturbance.
IMPORTANT SAFETY INFORMATION
Read MoreImportant Safety Information
SYNVISC and SYNVISC-ONE are contraindicated in patients with known hypersensitivity to hyaluronan products or patients with infections in or around the target knee. Hypersensitivity reactions including anaphylactic reaction, anaphylactoid reaction, anaphylactic shock and angioedema have been reported for both SYNVISC and SYNVISC-ONE.
Do not concomitantly use disinfectants containing quaternary ammonium salts for skin preparation because hyaluronan can precipitate in their presence. Do not inject SYNVISC or SYNVISC-ONE extra-articularly, into the synovial tissues, into the fat pad or joint capsule, or intravascularly. Some cases of skin necrosis have been reported after intra-articular use of hyaluronic acid. Patients should be instructed to contact their treating physician if signs of skin disorder (such as change of color or open sores) appear.
The safety and efficacy of SYNVISC-ONE in locations other than the knee, or for conditions other than osteoarthritis, or in combination with other intra-articular injectables, or in severely inflamed knee joints have not been established. Use caution when injecting SYNVISC or SYNVISC-ONE in patients allergic to avian proteins, feathers, or egg products; who have evidence of lymphatic or venous stasis in the leg to be treated; or who have severe inflammation in the knee to be treated. Remove any synovial fluid or effusion before injecting SYNVISC or SYNVISC-ONE. Strict adherence to aseptic technique must be followed to avoid joint infection. The safety and effectiveness of SYNVISC and SYNVISC-ONE have not been established in children (≤21 years old) or in pregnant or lactating women. Patients should be advised to avoid strenuous or prolonged weight-bearing activities for approximately 48 hours after treatment.
For SYNVISC
In clinical trials, the most commonly reported adverse events were transient pain, swelling, and/or joint effusion in the injected knee. The following reported adverse events are among those that may occur in association with intra-articular injections, including SYNVISC: arthralgia, joint stiffness, joint effusion, joint swelling, joint warmth, injection site pain, arthritis, arthropathy, and gait disturbance.
View the Complete Prescribing Information for SYNVISC
For SYNVISC-ONE
The most commonly reported adverse events were arthralgia, arthritis, arthropathy, injection site pain, and joint effusion. The following reported adverse events are among those that may occur in association with intra-articular injections, including SYNVISC-ONE: arthralgia, joint stiffness, joint effusion, joint swelling, joint warmth, injection site pain, arthritis, arthropathy, and gait disturbance.

